Suffering from musculoskeletal problems? You’re not alone.
Musculoskeletal problems are very common among orchestral musicians. There is still significant disagreement on the exact prevalence of these problems. Some studies, like the one conducted by Fotiadis and colleagues (2013), report 81.3% of the musicians they studied were affected, while others, like Ackermann and colleagues (2012), come back with 84%. The largest survey ever taken on orchestral musicians was conducted by the International Conference of Symphony Orchestra Musicians (ICSOM) in 1988. It revealed that 76% of the 2212 orchestral musicians surveyed had a medical problem that was severe enough to affect their playing. The neck and back were the most common sites for pain. (Fishbein, Middlestadt, Otatti, Straus, & Ellis, 1988). How does this happen? There are many potential causes, but commonly reported risk factors include personal factors like individual technique and posture, gender (women reportedly had higher rates of problems than men), age, type of instrument played (instrumentalists in strings, piano, harp, and percussion were found to be more vulnerable than the rest of the group), as well as occupational factors like number of hours played daily, perceived exertion of playing, and difficulties recovering after an episode of musculoskeletal pain related to training and playing load (Fotiadis et al., 2013; Kaakkola, Liira, Savolainen, Viljamaa, 2017).
What are some solutions to these problems?
Ackermann, Adams, & Marshall (2002) compared strength-training and endurance training in undergraduate music students with performance-related musculoskeletal disorders over a six week period. They conclude that while both groups showed positive results at the end of the six weeks, the endurance group had significant drops in perceived exertion of playing, suggesting that endurance training should be favored over strength training (Ackermann, Adams, & Marshall, 2002). From these results, Brandfonbrener (2003) concludes that “musicians need light resistance work with many repetitions rather than weight training” (Brandfonbrener, 2003, p. 237). She recommends swimming, calling it “an ideal and safe form of exercise for musicians” (p. 237). After the exertion is over, recovery begins. Because of orchestra musicians’ often-tight schedules as well as the differing demands of programs, Kaakkola and colleagues (2017) mention that recovery can be difficult. Thus, more research is needed into the post-concert recovery period. Before we can talk about recovery, we first need to know about some basic muscle structure and function.
Basic muscle structure and function
There are three different kinds of muscle tissue in the body: cardiac muscle – which is only found in the heart and that is involuntary, smooth muscle – which is found in the stomach, bladder, and intestines (among other places) and that is also involuntary, and skeletal muscle – which is voluntary (Marieb & Hoehn, 2013).
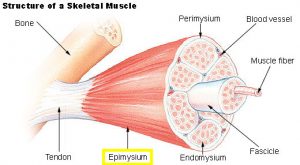
Each skeletal muscle is made up of multiple parts. Connective tissue sheaths cover and reinforce the different organizational units, stopping the muscle from bursting during very strong contractions. The first layer of dense connective tissue, found on the outside of the muscle, is called the epimysium (Figure 1). Within the muscle, bundles of muscle fibers are grouped into fascicles, which are surrounded by the perimysium, another layer of connective tissue. Finally, around each muscle fiber is a thin sheet of connective tissue called the endomysium. A single muscle fiber is made up of hundreds to thousands of myofibrils, or rod-like structures that contain the contractile elements of skeletal muscle, called sarcomeres. Sarcomeres are made up of alternating thick and thin filaments called myofilaments. The thick filaments are called myosin filaments, while the thin filaments are called actin filaments.
Figure 2. Myosin cross bridge showing calcium ions
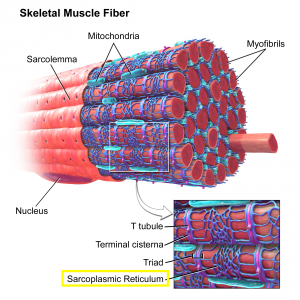
To execute a contraction, the nervous system must stimulate the muscle fiber. When a nerve impulse reaches the sarcoplasmic reticulum (SR) (Figure 3), the SR allows calcium ions to be released into the cytosol (the fluid inside of the cell), thus allowing the heads of the myosin filaments to bond to binding sites on the actin filaments. This binding forms a cross bridge (the link between the myosin head and actin) which allows the myosin heads to pull the actin filaments along, contracting the muscle (Figure 2) (Marieb & Hoehn, 2013).
What causes muscle fatigue?
According to Marieb and Hoehn (2013), muscle fatigue is a physiological inability of the muscle to contract even though it may still be receiving stimuli. Adenosine triphosphate (ATP) supplies the energy to form and detach the cross bridges. Although ATP availability declines during muscle contraction, it is very rare that muscles completely run out of ATP, thus Marieb and Hoehn conclude that it is not a factor in fatigue. When the nerve stimulates the muscle cell, potassium is lost from the cell and accumulates in the T tubules (the structure that allows the nerve impulse to reach the SR), which stops the release of calcium ions, interfering with contraction and leading to muscle fatigue.
But wait, I’ve always been taught that lactic acid causes muscle fatigue!
Although lactic acid was maligned for years as being a cause of muscle fatigue because it raises hydrogen ion concentration (which lowers the pH, making things more acidic) and alters contractile proteins, lactic acid actually counteracts high potassium levels, which do lead to muscle fatigue. Unless you are absolutely maximally exerting yourself, pH will be kept within normal limits (Marieb & Hoehn, 2013). Additionally, according to Westerblad and colleagues (2002), the concentration of inorganic phosphate determines the amount of muscle fatigue. The more inorganic phosphate that is present, the harder it is for the myosin heads to pull the actin strands along.
What are the differences between aerobic and anaerobic exercise?
Before we can talk about recovery, we have to talk about the difference between aerobic and anaerobic exercise. Adenosine triphosphate (ATP) is the most useful molecule in the body because it is used to power all functional processes, including muscle contraction. ATP is stored in the muscles for later use and has to be regenerated as fast as its broken down. This regeneration is carried out by three different methods (ordered from fastest to slowest): direct phosphorylation, the anaerobic pathway, and the aerobic pathway.[tabs]
[tab title=”Direct phosphorylation”]
- Function: Forms ATP by combining creatine phosphate (CP) – a unique high-energy molecule stored in the muscles – with adenosine diphosphate (ADP).
- Oxygen required: No
- Speed: Almost instant
- Energy source: CP
- Products: One ATP molecule per CP molecule.
- Energy provided: About fifteen seconds of maximal exertion – about as long as it takes to sprint 100 meters.
[/tab]
[tab title=”Anaerobic pathway”]
As the ATP and CP get used up, the body must switch to the anaerobic pathway.
- Function: ATP generated by breaking down glucose molecules into pyruvic acid (a product of the breakdown of carbohydrates)
- Oxygen required: No (anaerobic means “without oxygen”)
- Speed: Produces ATP about 2.5 times faster than aerobic respiration
- Energy source: Glucose
- Products: Two ATP molecules per one glucose molecule
- As muscles reach about 70% of their maximum possible activity, they compress their own blood vessels, interfering with blood flow and oxygen delivery. In this oxygenless environment, the pyruvic acid is converted to lactic acid which diffuses out into the bloodstream. It can then be picked up by the heart, liver, or kidneys and used as an energy source. Liver cells also have the ability to convert the lactic acid back into glucose or pyruvic acid and release it back into the bloodstream for muscle use.
- Energy provided: 30-40 seconds of ~ 70% maximal exertion (e.g. running 600 meters all-out)
[/tab]
[tab title=”Aerobic respiration”]
- Oxygen required: Yes
- Energy source: Glucose, pyruvic acid, amino acids from the breakdown of protein
- Speed: Slow
- Function: Complete breakdown of glucose molecule into carbon dioxide, water, and thirty two ATP
- Products: CO2, H2O, 32 ATP molecules per one glucose molecule
- Energy provided: Hours (given abundant oxygen and fuel from food) (Marieb & Hoehn, 2013).
[/tab]
[/tabs]
The effect of temperature on muscular recovery
According to Bishop and colleagues (2008), there are three types of recovery: immediate recovery (the recovery between strides of a runner where the muscle has to generate ATP and remove the byproducts of movement), short-term recovery (such as the sixty seconds or so between weightlifting sets or interval sprints), and training recovery (the period between successive workouts) (Bishop, Jones, & Woods, 2008).
Recovery is a contentious issue among athletes who are always looking for that slight edge over their opponents. Shorter recovery time means more time can be spent on training. But does it actually help recovery? To find out, Cheng and colleagues (2017) conducted a study where the participants performed three 5 minute all-out bouts of arm cycling exercise (on a machine that looks like you’re pedaling a bicycle but with your hands) to establish their baseline performance, followed by one hour of moderate intensity (50% of their maximum) exhaustive arm cycling runs. They were then given a two hour period to recover where both arms were, in the first group, heated to ~ 38ºC, in the second group, not heated (allowed to maintain body temperature ~ 33ºC), or, in the third group, cooled to 15ºC. Additionally, during the two hours, all subjects were told to continuously eat carbohydrates in the form of sports bars and drinks to maximize glycogen replenishment. When the two hours were up, participants performed the same three 5 minute all-out runs. Ultimately, the researchers found that heating helped maintain muscle power better than cooling because, due to the fact that chemical reactions slow down as they are cooled, glycogen resynthesis slowed. Also, prolonged exposure to cold temperatures causes blood vessels to constrict, which possibly further slowed the delivery of glycogen back to the muscles (Cheng et al., 2017).
I heard that cold water immersion after exercise reduces inflammation. Is this true?
Peake and colleagues (2017) conducted a study to hopefully shed some light on this question. Their subjects performed one bout of single-leg resistance exercise on two separate days (using alternate legs). On day one they would do the exercises using their right leg and on day two, they would do them with their left leg. The exercises were 45 degree leg press (six sets of 8-12 repetitions), single-leg squats (three sets of 12 reps), knee extensions (six sets of 8-12 reps), and walking lunges (three sets of 12 reps). Each session was followed with either cold water immersion (CWI) or active recovery. CWI began five minutes after the training session. Water was circulated at 10.3ºC with both legs submerged up to the waist for ten minutes. For the active recovery trial, participants cycled on a stationary bike at low intensity (producing ~ 36.6 watts) for ten minutes following their sessions. To assess changes in each of the participants, blood was drawn before the workout, immediately after the workout, immediately after the recovery therapies, and 30 min, 1h, 2h, 24h, and 48h after the workout. Muscle biopsies were collected before exercise, and 2h, 24h, and 48h after exercise. After it was all said and done, the researchers found no significant differences in inflammation between active recovery and CWI (Peake et al., 2017).
Are there any benefits associated with CWI?
The short answer is that, in the short term, there might be. Stanley and colleagues (2012) found perceived improvement of muscle soreness and fatigue (Stanley, Buchheit, & Peake, 2012), while Roberts and colleagues (2014) found enhanced recovery of submaximal muscle performance following resistance training (Roberts et al., 2014). Despite these findings, McPhee and Lightfoot (2017) note that the efficacy of CWI therapy largely depends on the type of exercise being undertaken and that more research is needed to comprehensively address the impact of post-exercise CWI (McPhee & Lightfoot, 2017). CWI might help your perception of recovery by altering either blood flow and pressure inside the muscle, or activity of pain receptors in the muscle (Stanley, Buchheit, & Peake, 2012).
If you’re going for strength gains, according to Roberts and colleagues (2015), CWI could inhibit protein synthesis, thus leading to less improvement in muscular size and strength (Roberts et al., 2015). If you have any other injury, however, physiotherapists still recommend RICE (Rest, Ice, Compression, Elevation) as a first line of defense (Hambleton, 2013).
Some takeaways
Any exercise is good for you and should be done on a regular basis, but swimming is a particularly good one for musicians. There is no one-size-fits-all approach to recovery. There have been many studies done on the benefits cold water immersion confers; the conclusion drawn is that cold water immersion has advantages and disadvantages depending the type of exercise you do. Since playing an instrument is an endurance exercise, cold water immersion likely will not help your muscular recovery; you are better off heating the muscles. However, it might help your perception of recovery, which, in a psychologically high-stress field like music, is a good thing. If you are looking for a way to reduce muscle inflammation following strength training, no difference between cold water immersion and active recovery has been found. Cold water immersion following resistance training might impact your strength gains, so it’s best to stick with active recovery. It might have a positive effect on submaximal muscle performance following strength training. So, if you would like to do cold water immersion following particularly difficult or long periods of playing, go for it. It might even enhance feelings of recovery. But, there’s no evidence of physiological improvement in endurance exercise like playing an instrument. If you have tendon pain, remember: RICE until you can see a physiotherapist.
[toggle title=”References”]
Articles
Ackermann, B., Adams, R., & Marshall, E. (2002). Strength or endurance training for undergraduate music majors at a university? Medical Problems of Performing Artists, 17(1), 33-41. Retrieved from https://www.sciandmed.com/mppa/journalviewer.aspx?issue=1085&article=953.
Ackermann, B., Driscoll, T., & Kenny, D.T. (2012). Musculoskeletal pain and injury in professional orchestral musicians in Australia. Medical Problems of Performing Artists, 27(4), 181-187. Retrieved from https://www.sciandmed.com/mppa/journalviewer.aspx?issue=-1&year=2012.
Bishop, P.A., Jones, E., & Woods, A.K. (2008). Recovery from training: a brief review. Journal of Strength and Conditioning Research, 22(3), 1015-1024. https://doi.org/10.1519/JSC.0b013e31816eb518.
Brandfonbrener, A. (2003). Musculoskeletal problems of instrumental musicians. Hand Clinics, 19(2), 231-239. https://doi.org/10.1016/S0749-0712(02)00100-2.
Cheng, A., Willis, S.J., Zinner, C., Chaillou, T., Ivarsson, N., Ørtenblad, N.,… Westerblad, H. (2017). Post-exercise recovery of contractile function and endurance in humans and mice is accelerated by heating and slowed by cooling skeletal muscle. The Journal of Physiology 595(24), 7413-7426. https://doi.org/10.1113/JP274870.
Fishbein, M., Middlestadt, S.E., Ottati, V., Straus, S., & Ellis, A. (1988). Medical problems among ICSOM musicians: An overview of a national survey. Medical Problems of Performing Artists, 3(1), 1-8. Retrieved from: https://www.sciandmed.com/mppa/journalviewer.aspx?issue=1145&article=1451.
Fotiadis, D.G., Fotiadou, E. G., Kokaridas, D.G., & Mylonas, A.C. (2013). Prevalence of musculoskeletal disorders in professional symphony orchestra musicians in Greece. Medical Problems of Performing Artists, 28(2), 91-95. Retrieved from: https://www.sciandmed.com/mppa/journalviewer.aspx?issue=-1&year=2013.
McPhee, J.S. & Lightfoot, A.P. (2017). Post-exercise recovery regimes: blowing hot and cold. The Journal of Physiology, 595(3), 627-628. https://doi.org/10.1113/JP273503.
Hambleton, L. (2013). Pro athletes recover faster than amateurs partly because they get superior medical care. The Washington Post. Retrieved from: https://www.washingtonpost.com/national/health-science/pro-athletes-recover-faster-than-amateurs-partly-because-they-get-superior-medical-care/2013/11/11/9da3385c-d291-11e2-8cbe-1bcbee06f8f8_story.html?utm_term=.dc2384b53aec.
Kaakkola, S., Liira, J., Savolainen, A., & Viljamaa, K. (2017). Musculoskeletal symptoms among Finnish professional orchestra musicians. Medical Problems of Performing Artists, 32(4), 195-209. Retrieved from: https://www.sciandmed.com/mppa/journalviewer.aspx?issue=1219&article=2208.
Peake, J.M., Roberts, L.A., Figueiredo, V.C., Egner, I., Krog, S., Aas, S.N., … Raastad, T. (2017). The effects of cold water immersion and active recovery on inflammation and cell stress responses in human skeletal muscle after resistance exercise. The Journal of Physiology, 595(3), 695-711. https://doi.org/10.1113/JP272881.
Roberts, L.A., Nosaka, K., Coombes, J.S., & Peake, J.M. (2014). Cold water immersion enhances recovery of submaximal muscle function after resistance exercise. American Journal of Physiology – Regulatory, Integrative and Comparative Physiology, 307(8), R998-R1008. https://doi.org/10.1152/ajpregu.00180.2014.
Roberts, L.A., Raastad, T., Markworth, J.F., Figueiredo, V.C., Egner, I.M., Shield, A. … Peake, J.M. (2015). Post-exercise cold water immersion attenuates acute anabolic signalling and long-term adaptations in muscle to strength training. The Journal of Physiology, 593(18), 4285-4301. https://doi.org/10.1113/JP270570.
Stanley J., Buchheit M., & Peake, J.M. (2012). The effect of post-exercise hydrotherapy on subsequent exercise performance and heart rate variability. European Journal of Applied Physiology 112(3), 951–961. https://doi.org/10.1007/s00421-011-2052-7.
Westerblad, H., Allen, D.G., Lӓnnergren, J. (2002). Muscle fatigue: lactic acid or inorganic phosphate the major cause? Physiology, The American Physiological Society, 17(1), 17-21. https://doi.org/10.1152/physiologyonline.2002.17.1.17.
Books
Marieb, E.N., Hoehn, K. (2013). Human anatomy and physiology. (n.p.): Pearson Education.
Images
Figure 1. Illu muscle structure, by Arcadian, 2006. Public domain.
Figure 2. 1008 Skeletal Muscle Contraction, by Open Stax, 2016. CC by 4.0.
Figure 3. Blausen 0801 SkeletalMuscle, BruceBlaus, 2013. CC by 3.0.
[/toggle]
Author: Sam Shreves

Excellent , very in depth article , which addresses a common issue for musicians . Educational and enlightening! Thank you! Lauren Spencer
Very interesting and comprehensive.
Yes, I’m surely believe that there is pain associated with continued movement. I once had tendinitis in my left elbow from gardening! After physical therapy,, it continued to hurt. After about 2 years it just resolved itself. Who knew?
Interesting & informative. I’ve offended wondered how musicians play their instrument in rapid succession without having to stop for a break!
Hi Vicki (Mom), thanks so much for reading! Well, we often play even though we’re tired or it hurts. It’s not a good practice (hah) and hopefully these blogs help people understand that playing through the pain is really not advisable. – Sam